The 4 Primary Reasons We Age and What To Do About It, Part 1

It is not settled science, but there may be just four primary reasons we age that instigate several reactions, which in total explain the aging process. Here I summarize the primary reasons our body and mind degrades over time and what can be done about it.
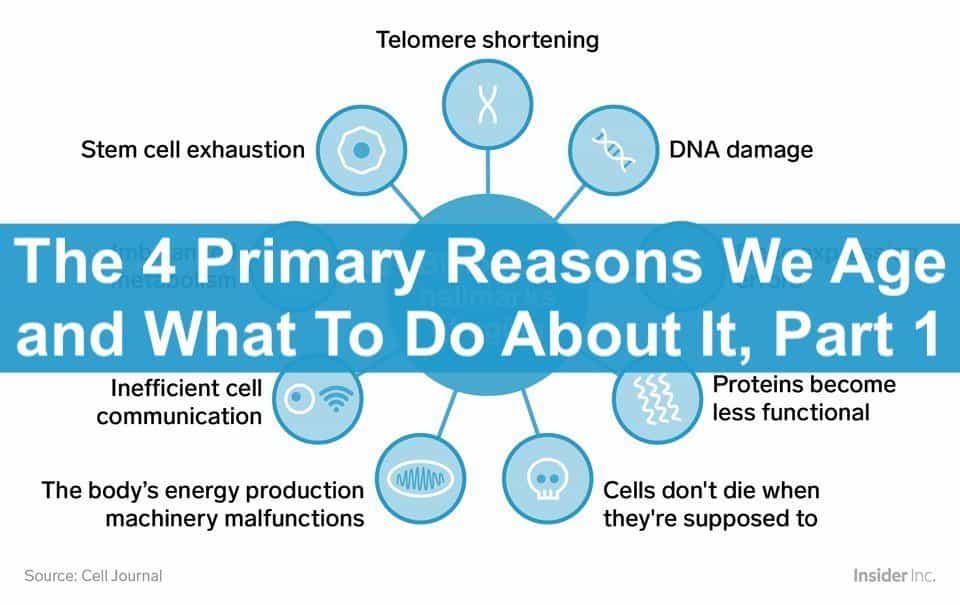
IN 2013, a group of Spanish scientists led by Dr. Carlos Lopez-Otin took a crack at identifying and explaining what they referred to as the “Hallmarks of Aging”. Since then, their study has become one of the most referenced studies in the aging science literature.
In this piece, I’m going to explain the conclusions of the Hallmarks of Aging study and suggest specific interventions (such as exercise, supplements and even a drug) that you can do to slow down how fast you age, and possibly reverse some of the aging-instigated damage you now experience.
These are the nine so-called “Hallmarks of Aging”:
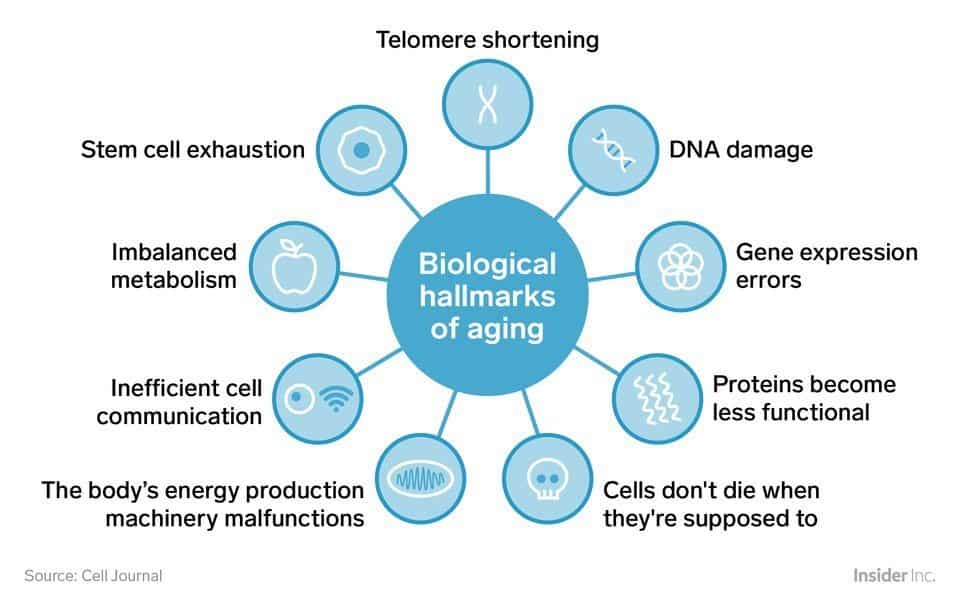
That’s a nice, clean illustration, but it doesn’t show some cause and effect. In fact, there’s not yet a widely accepted model proving the precise reasons we age and why and how the observable physical and cognitive degradation are connected.
Of the nine “hallmarks of aging”, here in Part 1, the focus is on the “Primary” root causes of aging, as defined by the Hallmarks study. In Part 2, we’ll chew on the “Compensatory” and “Integrative” hallmarks of aging, which pretty much means how our body’s biological systems respond to and compensate for the four Primary causes.
Here’s what we’re going to cover:
- How ordinary DNA damage becomes an extraordinary problem when our bodies can no longer fix it.
- What progressively shortening telomeres have to do with aging and lifespan.
- The beauty of epigenetics — yes, you can influence turning “off” bad and turning “on” good genes.
- Why protein is much more than a muscle-feeder, given it must function properly for us to age well.
In each section, I’ll explain the science a bit and what interventions are available for you to adopt to improve the quality of your healthspan.
Let’s dig in…
Summary of the Hallmarks of Aging
The common characteristic of the Primary hallmarks of aging is the fact that they are all unequivocally negative. This is the case with:
- DNA damage (including during cell division when chromosomes do not separate properly between the two cells (chromosomal aneuploidies)), mitochondrial DNA mutations and telomere loss (they get shorter);
- Epigenetic drift, (altered epigenome due to aging); and
- Defective proteostasis (dysfunctional proteins).
As you’ll soon learn below and in Part 2, in response to the four primary hallmarks of aging are three “Antagonistic” hallmarks that attempt to compensate for the damage done by the first four, and two “Integrative” hallmarks that arise when the accumulated damage caused by the Primary and Antagonistic hallmarks cannot be compensated by tissue homeostatic mechanisms (body cell/tissue regulation).
The next illustration comes from The Hallmark of Aging study. The illustration above already named the nine hallmarks of aging identified by Dr. Lopez-Otin et al, but what’s next will give you some sense of how they relate to one another.
- “Primary” hallmarks are the root or primary causes of aging (covered in this piece):
- Genomic instability (DNA damage)
- Telomere attrition (chromosome “caps” become less protective)
- Eipgenetic alterations (gene expression compromised)
- Loss of proteostasis (proteins are damaged)
- “Antagonistic” hallmarks are compensatory responses to the primary ones that initially mitigate the damage made by them, but if chronic or exacerbated, these antagonistic hallmarks become deleterious themselves (covered in Part 2):
- Deregulated nutrient sensing (imbalanced metabolism)
- Mitochondrial dysfunction (faltering energy)
- Cellular senescence (zombie cells)
- “Integrative” hallmarks are the result of the previous two groups of hallmarks and are ultimately responsible for the functional decline associated with aging (covered in Part 2):
- Stem cell exhaustion (tissues no longer get repaired)
- Altered cellular communication (cell “communication” compromised)
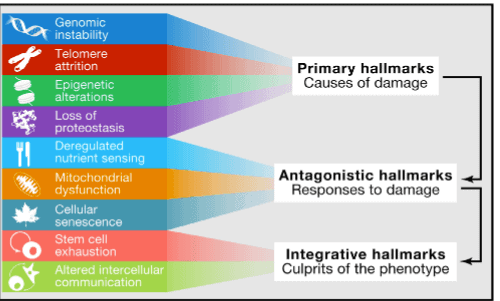
This is Figure 6 from The Hallmarks of Aging study
Next, let’s look at each Primary aging “hallmark” and show which interventions may be useful to ameliorate the negative impact they have in making us old, frail and susceptible to chronic disease.
The 4 Primary Hallmarks of Aging
As mentioned, they are:
- Genomic instability, which results in DNA damage;
- Telomere attrition, the shortening of protective chromosomal “caps”;
- Epigenetic alterations, which happens when gene expression is compromised; and
- Loss of Proteostasis, which happens when proteins become less stable and accurate.
1. Genomic instability/DNA damage (Primary)
What this means: Errors appear in DNA that if not correct degrade various physical and mental functions.
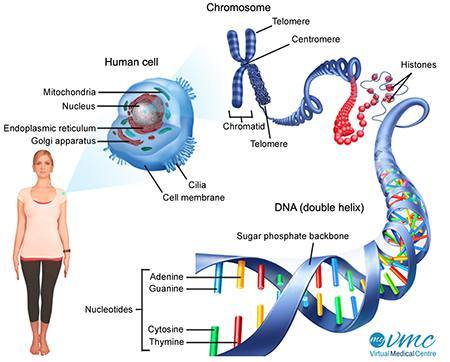
What’s DNA? Deoxyribonucleic acid (DNA) is a molecule composed of two chains that coil around each other to form a double helix carrying the genetic instructions used in the growth, development, functioning and reproduction of all known living organisms and many viruses. DNA is made out of two long, twisted strands that contain complementary genetic information (like a picture and its negative). A gene is a segment of DNA that is passed down from parents to children and confers a trait to the offspring. Genes are organized and packaged in units called “chromosomes.” (1)
What you need to know — DNA carries genetic instructions for all the body’s functions that are visualized as a succession of letters (A,G,C,T) that show the order of nucleotides, but it’s not infallible. As it replicates, errors happen, and the mechanisms that exit to correct them gradually become less effective as we age.
Every time a human cell divides and its DNA replicates, it has to copy and send the same sequence of 3 billion nucleotides (the basic structural unit of nucleic acids such as DNA ) to its daughter cells. Each of our cells takes a DNA damage hit at the rate of 10,000 to 1,000,000 molecular lesions per cell per day, and thus genome instability is considered the first of the primary reasons we age by the Hallmarks of Aging study.
Mistakes happen; in fact “errors” are a natural part of DNA replication. For instance, over a lifetime a person may rack up more than 2,000 DNA mutations per brain cell. (2)
One type of damage that occurs with increasing frequency as we age is that the DNA code might not be correctly copied during replication. Parts could get misspelled and sections could be accidentally inserted or deleted. These errors are not always caught by the mechanisms in our bodies that repair DNA, which include a family of enzymes called glycosylases. (3)
Age isn’t the only thing that can inhibit the repair of DNA damage. Lifestyle choices that cause psychological and physiological stress can also hinder DNA repair, including disrupted sleep patterns, smoking or alcoholism. Some scientists suspect that yet to be discovered mechanisms contribute to failing DNA repair, such as an epigenetic clock, popularized by UCLA professor Steve Horvath and his Horvath Aging Clock.
What You Can Do
To restore the body’s DNA repair mechanisms, Drs. Leonard Guarente and David Sinclair, among others, discovered that boosting levels of NAD helps keep up DNA repair in mice. Dr. Guarante’s company, Elysium Health, sponsored human trials that showed NAD levels to dramatically improve by taking an NAD precursor called NR. Dr. Sinclair is conducting human trials to show the same with another NAD precursor called NMN, after his studies with mice showed remarkable anti-aging effects of the compound.
For more on this read Can NAD+ Precursors NR and NMN Make You Young Again?
2. Telomere attrition (Primary)
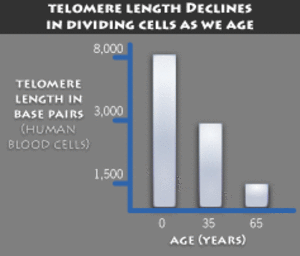
What this means: As cells divide the “caps” at the end of each chromosome tend to get shorter, and as such some scientists believe telomere length to be a nearly precise marker for lifespan.
What are telomeres? They’re repetitive nucleotide sequences at each end of a chromosome, which protects the end of the chromosome from deterioration or from fusion with neighboring chromosomes. (4)
What you need to know — Telomeres protect each strand of DNA. They’re often compared to the plastic tips of shoelaces that keep them from fraying. As they get shorter with each cell division, chromosomes can become unstable and have correct replication problems. And enzyme called Telomerase may be able to extend the length of telomeres and thereby extend lifespan.
Telomeres are repeated patches of DNA that keep up the stability of our chromosomes and protect our genetic data, and are considered by the Hallmarks of Aging study to be the second of the primary reasons we age.
In addition to protecting genes during cell division, telomeres act as a biological clock in our cells. Each time a cell divides, its telomeres get shorter, until the cell either undergoes a form of cell death called apoptosis or becomes senescent.
DNA becomes vulnerable to degradation when telomeres wear away, and thereby become gradually less and less protective, whether because of aging or environmental stress (meaning, stress exogenous to — outside — the body). Shorter telomeres cause genomic instability and may fuel cancer if it’s already present.
To repair telomeres some of our cells use an enzyme called telomerase to rebuild the shortened ends. Studies with mice show telomere elongation via telomerase can increase their lifespan.
What You Can Do
Geroscientists are looking at ways they can lengthen our telomeres to lead longer, healthier lives. Using RNA therapy to lengthen telomeres, one doctor was able to extend the lifespan of human cells in culture.
The only commercially available potential intervention is a agglomerate-activating supplement called TA-65 made from astragalus, a Chinese herb, and other proprietary ingredients. I would recommend if it wasn’t so expensive, and had the potential to amp up the proliferation of cancer cells, a potential result of augmenting telomeres. Perhaps it would be safer to consume the less potent astragalus herb itself.
Read my article, Three Months to Longer Life.
3. Epigenetic alterations — Gene expression gets compromised (Primary)
What this means: Genes that are harmful to long-term health get expressed (“turned on”).
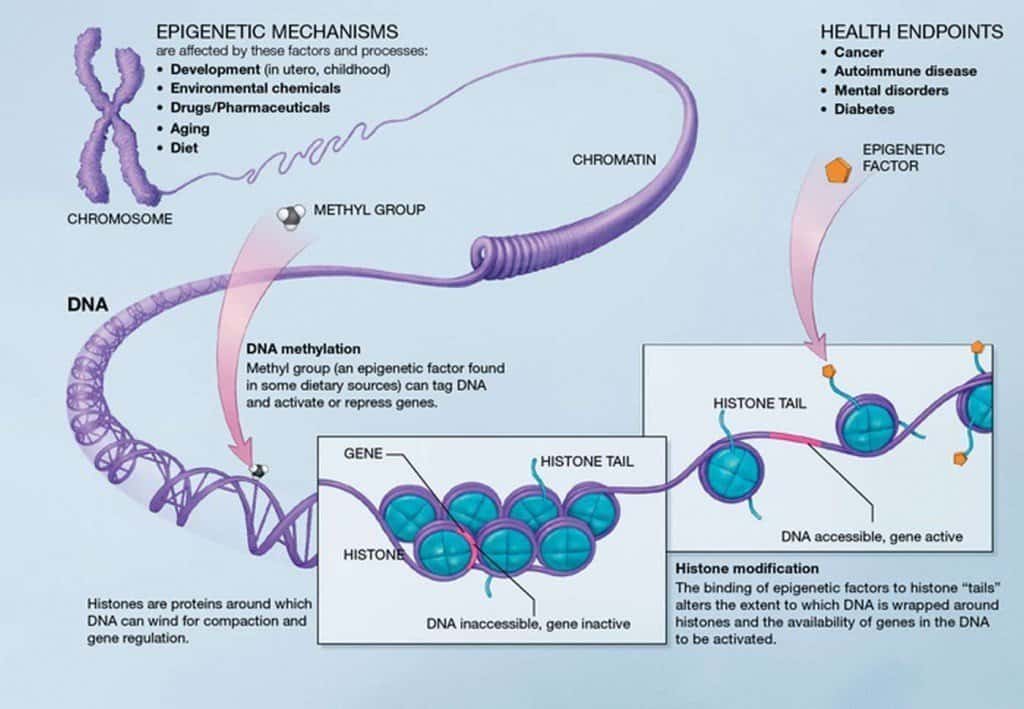
What’s Epigenetics? It’s the study of changes in organisms caused by a change of gene expression and not alteration of the genetic code itself. Epigenetics is the process of dictating which DNA segments are expressed.
What you need to know — The DNA that gets “read” and thereby translated into physical traits become compromised as we age. For example, if an inadvertent change results in the silencing of a gene that helps suppress tumors, cells could uncontrollably grow into cancer.
(Credit: NIH.gov)
Epigenetic alternation is considered the third of the primary reasons we age. It’s different that DNA damage per se, because epigenetics isn’t about the replication of DNA code, but how that code gets expressed physiologically via gene expression.
As we age, the proteins bound to DNA become looser and less exact, and genes start to get expressed when they shouldn’t be, or get silenced in error. This means that some necessary proteins aren’t being made, and some harmful, unnecessary proteins are.
The epigenome is the master program which controls our genetic code. An epigenetic program makes a series of temporary changes to our genome, and runs the show by turning genes off or on, and controlling protein production in particular cells.
Our environment influences our epigenome and modifies it via epigenetic changes. As we age, our cells are continually under assault by toxins and other stressors which change the epigenome. These changes accumulate over time, and geroscientists have linked them to the dysfunction and decline we associate with aging. Epigenetic alterations can occur via changes in RNA interference, histone modifications, and more commonly, DNA methylation.
Fortunately, changes in the epigenome are not permanent, and theoretically, this hallmark of aging can be reversed. As Dr. Carlos López-Otin says in his Hallmarks of Aging:
Unlike DNA mutations, epigenetic alterations are – at least theoretically – reversible, hence offering opportunities for the design of novel anti-aging treatments.
What You Can Do
Since the publication of The Hallmarks of Aging in 2013, scientists have made significant progress in understanding the epigenome, but little progress developing potential drug therapies. The changeability of the epigenome, as well as the recent discovery of epigenetic clocks, makes this hallmark of aging a potentially target for drug interventions.
Notwithstanding the absence of beneficial drugs, there are things you can do to help ensure the favorable alternation of your genes, such as:
- Meditation and yoga turn down the expression of pro-inflammatory genes thus reducing inflammation. Chronic inflammation is an underlying cause of seven of the top ten leading causes of death including cancer, heart disease, diabetes, and Alzheimer’s. (5, 6)
- Broccoli (particularly sprouts), green tea, cauliflower, kale, and other plant-based foods have a positive effect on gene expression. (7,8,9)
- Exercise, particular working out large muscles like the thighs. (10)
Read my articles:
Meditate Like A Monk In 20 Minutes
4. Loss of Proteostasis — Proteins become less stable and accurate in their roles (Primary)
What this means: Proteins perform integral functions in the cell. If they become compromised, as they tend to do as we age, then the ability of cells to clean out toxic waste and repair themselves is compromised.
What’s Proteostasis? The word combines “protein” and “homeostasis“, and refers to the competing and integrated biological pathways within cells that control the biogenesis, folding, trafficking and degradation of proteins present within and outside the cell. (11)
What you need to know — Proteostasis is central to understanding the cause of diseases associated with excessive protein misfolding and degradation leading to loss-of-function of various physical and mental attributes, including Alzheimer’s disease.
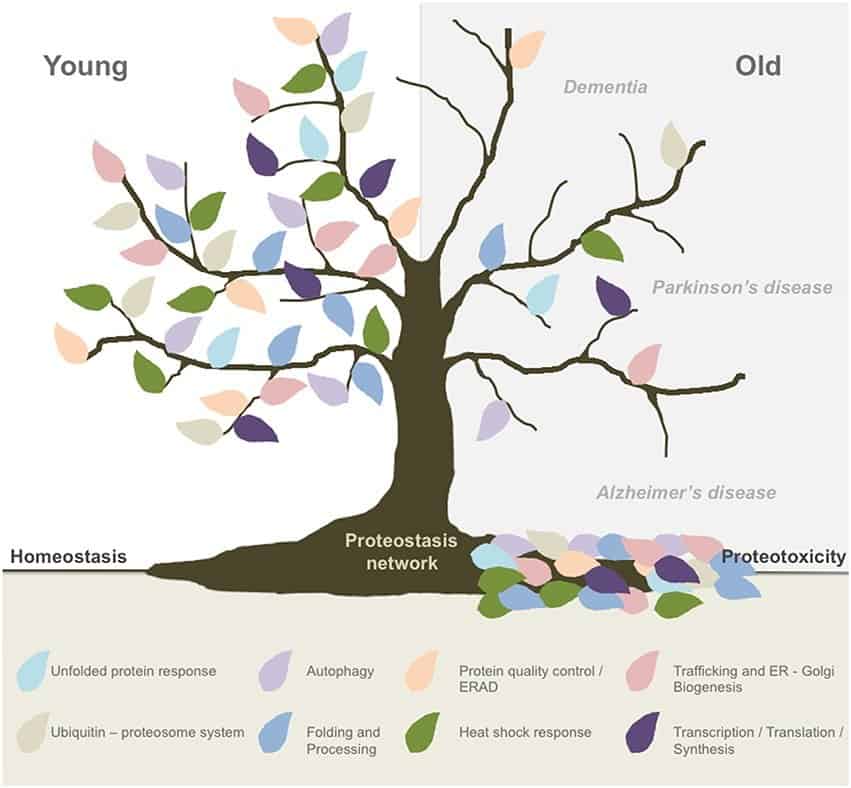
The decline in the protein quality of our cells, called the loss of proteostasis, is a fundamental hallmark of aging, and considered the fourth and last of the primary reasons we age, as defined by the Hallmarks of Aging study.
Our bodies have defenses against cellular stress; however, after decades of repeated assaults by stressors such as free radicals and other toxins, the proteins in our cells become damaged or misfolded.
Toxins are a significant source of disease and aging. For example, researchers have shown that free radicals in polluted air are particularly toxic, contributing to multiple age-related diseases. A major study published in the Lancet medical journal attributes one of out every six premature deaths worldwide in 2015 to disease resulting from exposure to environmental toxins in the air and water.
In younger cells, our autophagy housekeeping system clears out these unfolded proteins. In aging cells, unfortunately, this vital process degrades and the lysosomes become less efficient at eliminating this cellular toxic waste.
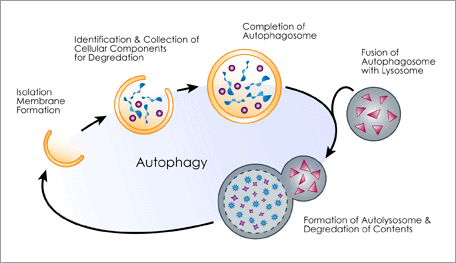
Geroscientists consider the decline in cellular autophagy a significant contributor to disease and aging, making the loss of proteostasis a primary hallmark of aging. The damage accumulates over the decades, and the mechanisms responsible for maintaining proper protein composition begin to decline. Proteins lose their stability, autophagy quality-control processes start to fail, and misfolded proteins accumulate, in a process known as cellular garb-aging.
Misfolded proteins form aggregates which sometimes produce disease. Amyloid proteins are an example of such aggregates, and play a prominent role in neurological conditions such as Huntington’s, Alzheimer’s, and Parkinson’s disease.
Chronic inflammation is also a byproduct of misfolded proteins. In a vicious cycle known as inflammaging, this build-up of cellular garbage increases the production of pro-inflammatory chemicals in our tissues which generates chronic inflammation throughout the body. This chronic, low-grade inflammation further accelerates the aging process.
What You Can Do
Maintaining protein quality control and active cellular autophagy is essential to a long and vibrant healthspan. To do this, studies show that it’s essential that you keep up/build muscle, as well as take regular saunas and practice some form of Intermittent Fasting. (12, 13, 14)
Read my articles:
The Functionally Fast Fit Workout
8 Ways Sauna-induced Hyperthermic Conditioning Can Help You Live Long and Strong
Your Takeaway
Here in Part 1 we covered the four primary reasons we age as defined by a landmark study headed by Dr. Carlos Lopez-Otin called, The Hallmarks of Aging.
The first three of the four primary reasons we age concern DNA and genes. Recall that a gene is a segment of DNA passed down from parents to children thereby conferring a trait to offspring. These genes are organized and packaged as chromosomes.
Thus, DNA damage, telomere shortening and epigenetics (which genes turn “on” and “off”) all start with DNA.
The fourth primary reason we age has to do with the stability and accuracy of proteins, an essential part of all living organisms, especially as structural components of body tissues such as muscle, hair, collagen, etc., and as enzymes and antibodies. Excessive protein misfolding and degradation is a primary cause of disease.
All of these four primary reasons we age get amplified as we age; therefore, to age better, age slower, age without the chronic diseases associated with aging, we must find ways to intervene, thwart and retard the mechanisms behind aging.
So there you have it, the four primary reasons we age, and what you can do to slow it all down. Next week in Part 2, I’ll dig into the three compensatory responses and two phenotype effects that result from the unfortunate assault we call “aging”.
Last Updated on February 7, 2024 by Joe Garma










