Can The Right Gut Bacteria Fight Obesity And Slay Metabolic Syndrome? (Part 1/2)

Yes, leptin and insulin do play a part in making us fat and sick, and could lead to metabolic syndrome, but inflammation and the two pounds of microbiota in our gut may play a more critical role than ever before imagined.
DO YOU have any idea what happens within minutes of eating a McDonald’s breakfast of an egg sandwich with cheese and ham, a sausage muffin sandwich, and two hash brown patties?
Besides a few burps?
Dr. Paresh Dandona, a diabetes specialist in Buffalo, New York, wanted to know. As scientist-types do, he conducted an experiment to find out.
Over several mornings, he fed nine normal-weight volunteers that eggie, chessey McDonald’s breakfast, which may seem an unhealthy thing to do to some of you, but for much of America (and the world at large), it’s typical of the high-fat, high-carbohydrate meals that 25% of Americans regularly eat.
What did Dr. Dandona discover?
He was shocked to find that levels of a C-reactive protein — an indicator of systemic inflammation — dramatically surged within minutes.
And the inflammation lasted for hours. Take a look:
Why care about inflammation? To answer that question, let’s examine “metabolic syndrome”.
Metabolic Syndrome: A Club You Don’t Wanna Join
Metabolic Syndrome is the name for a group of risk factors that raises your risk for a cluster of dysfunctions that tend to precede full-blown diabetes, and that also increase the risk of heart disease, stroke, certain cancers, and even dementia—the top killers of the developed world.
It’s not an unobtrusive syndrome. Having just three of the following five conditions are risk factors that place you in the quickly growing, multi-million member Metabolic Syndrome Club:
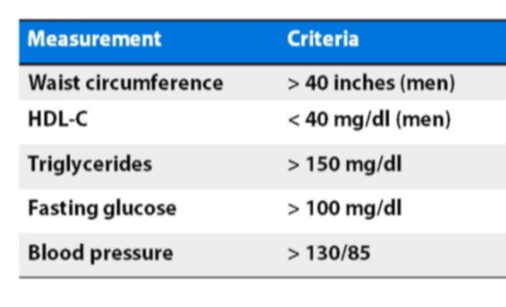
1. A large waistline. This also is called abdominal obesity or “having an apple shape.” Excess fat in the stomach area is a greater risk factor for heart disease than excess fat in other parts of the body, such as on the hips.
2. A high triglyceride level. Triglycerides are a type of fat found in the blood.
3. A low HDL cholesterol level. HDL sometimes is called “good” cholesterol because it helps remove cholesterol from your arteries. A low HDL cholesterol level raises your risk for heart disease.
4. High blood pressure. Blood pressure is the force of blood pushing against the walls of your arteries as your heart pumps blood. If this pressure rises and stays high over time, it can damage your heart and lead to plaque buildup.
5. High fasting blood sugar. Even mildly high blood sugar may be an early sign of diabetes.
More specifically, in a presentation given in June of 2013 at the Institute of Human and Machine Cognition, called, An Advantaged Metabolic State: Human Performance, Resilience & Health, Dr. Peter Attia offered these upper limits on waist circumference, HDL, triglycerides, fasting glucose and blood sugar:
If you know your numbers, you might wish to compare them to the criteria in the above chart. (To hear Dr. Attia’s discussion on this topic, go to the 5:27 mark of his video presentation here.)
The reason we care about inflammation is that if persistent it has a pernicious role in contributing to, or even causing, each of the five risk factors for metabolic syndrome listed above, which are antecedent to the leading causes of death in America.
While it’s true that inflammation is essential to help combat the microbes in an infected laceration or a virus causing the flu, when it goes on for too long (persistent/chronic), inflammation can become a key influence in metabolic syndrome-type damage.
That chronic, low-grade inflammation has long been recognized as a feature of metabolic syndrome is without dispute, but recently, doctors have begun to question whether chronic inflammation is more than just a symptom of metabolic syndrome: Could it, in fact, be a major cause?
The proposition that inflammation could be a significant cause of metabolic syndrome became the null hypothesis that Dr. Dandona sought to disprove. He spent a decade testing the effects of various foods on the immune system. His breakthrough moment came when he discovered that certain foods with near identical characteristics could have a completely different effect on the body due to something quite small and oft neglected.
Those Pesky Endotoxins
Orange juice provided the revelation.
Nutritionists warn parents not to replace water with fruit juice for their children because of all the sugar contained in fruit juice. This is good advice, but interestingly, orange juice contains something that shield’s the metabolism from the deleterious effects of inflammation and elevated blood sugar. (At least in modest amounts consumed.)
As reported in Mother Jones magazine, upon which much of this blog post is based, Dr. Dandona observed this phenomenon in a test with ten volunteers. He gave them all a McDonald’s type breakfast, but each of three groups drank something different:
One group quaffed orange juice.
One group gulped sugar water.
One group guzzled plain water.
All but the orange juice-drinking group experienced elevated blood sugar and inflammation.
Why?
It’s the endotoxins, naturally.
(OK, I’ve barely heard about it myself.)
Endotoxin is a molecule that comes from the outer walls of certain bacteria. When endotoxin levels rise, our immune system perceives a threat and responds with inflammation. This is what happened with the MacDonald-eating-non-OJ group.
Where did the endotoxins come from?
There are two possibilities:
– The endotoxins were in the food consumed, or
– The endotoxins came from the test subject’s guts.
As I wrote in How To Feed A Trillion Guests:
“There’s more to you than you think. Like 100 trillion more. Your gut bacteria have an enormous influence on your health.”
Although they exist everywhere upon and in us, most of the trillions of bacteria we have lives in our gut, about two pounds worth. (Yeah, they’re tiny, skinny creatures.)
Collectively, they’re referred to as “microbiota”.
Within that microbiotic cauldron live endotoxins. Dr. Dandona and others of his ilk think that greasy, refined carbohydrate-filled food attracts it from its harmless residence in the gut into the bloodstream where it elevates blood sugar and inflammation.
But this did not happen with the orange-drinking group that Dr, Dandona studied, despite their consumption of greasy, fatty food.
Why?
Somehow, the orange juice stopped the translocation of the endotoxins from the gut, where they do no harm, into the blood stream where plenty of harm ensues, which, if true, suggests that the movement of inflammation-producing endotoxins in the gut into the blood stream is more detrimental than those that may already be present in food.
What in our body has the following attributes?
- Having 150 times more genes, it represents 90% of our physical form.
- Is represented by between 500 and 1,000 species.
- Containing as many neurons as the spinal cord, it has its own nervous system.
- They enable our immune and digestive systems to function.
Well, by now you may guess the answer… it’s our microbiota, which some scientists are now calling, our “forgotten organ”.
And it may hold the key to addressing our single greatest health threat: “diabesity”, as Dr. Mark Hyman calls this frequent collaboration between obesity and diabetes.
Currently, one-third of Americans are obese and another one-third overweight. Go randomly grab three people from a shopping mall, and two will be oversized.
This is a problem.
As the aforementioned Mother Jones reports:
“In 2011, the United Nations announced that for the first time ever, chronic diseases, most of which are linked to obesity, killed more people than infectious diseases.” (Source)
Why is obesity so prevalent?
The conventional wisdom says that the obese eat too much of the wrong things and exercise too little.
That appears to makes sense.
Given that a pound of body fat contains about 3,500 calories, if someone routinely eats 500 calories more than expended – say, one bagel lavished with cream cheese – then it’s expected that one extra pound of fat will accumulate on his body each week (7 x 500).
But the truth is, not everyone who eats such a surplus of calories gains the same amount of weight during the same amount of time. In fact, some fat people have well functioning metabolisms, whereby their insulin and blood sugar numbers are healthy, and their livers are not marbled with fat.
Surprisingly, some thin people are just the opposite of what you’d expect. They too can have metabolic syndrome. One clue is someone who appears thin everywhere but has a beer belly. Such a person could suffer from fatty liver, insulin resistance, elevated blood sugar, high blood pressure, and systemic inflammation.
Whether thin or fat, the common thinking among health scientists is that someone joins the Metabolic Syndrome Club in this routine, specific, eight-step cascade:
- You eat too much greasy, highly refined food.
- Calories flood your body.
- The insulin that would ordinarily help your cells absorb these calories is overwhelmed by the concentration of caloric energy (sugar) and the cells become unresponsive.
- Your pancreas kicks in to help out and begins producing more insulin.
- Once your pancreas finally shuts down from exhaustion, you get diabetes.
- What’s more, you develop resistance to another hormone called leptin, which signals satiety (fullness).
- With leptin impaired, you tend to overeat.
- Meanwhile, fat cells — which have become bloated and stressed as they try to store the excess calories — begin producing low-grade inflammation.
As mentioned, the above eight-step process is the prevalent thinking about how metabolic syndrome happens. There is, however, new research being done that offers another scenario.
Inflammation might not be a symptom – the thing that happened in step number eight – but the cause of metabolic syndrome, the precursor of all those nasty, deadly diseases that smite so many of us.
Here’s how the inflammation theory goes, summarized in this four step cascade:
- You eat some greasy, highly refined food.
- Your immune system is activated (via an inflammation response) by this food, which prompts insulin and leptin resistance.
- Sugar (glucose) builds up in your blood, which causes more insulin to be produced.
- Your pancreas (producing the insulin) and liver (filtering the blood) are overwhelmed.
All this happened because inflammation cripples your cells’ ability to adequately respond to hormonal signals (insulin and leptin).
An important piece of supportive evidence of this theory is from experiments with obese mice with metabolic syndrome. By simply increasing the number of T-cells (the white blood cells that reduce inflammation), researchers discovered that the mice’s metabolic syndrome faded away.
Nothing else was done to reverse the metabolic syndrome. Stop the inflammation; end the syndrome.
What Have We Learned So Far?
Here are the key takeaways from this exposition on metabolic syndrome, microbiota and inflammation:
- Metabolic syndrome is a nasty condition that you want to avoid, because it dramatically increases your chances for an unhappy life and untimely death.
- If you have any three of five risk factors for metabolic syndrome described above, you’ve joined that unhappy club.
- Microbiota comprise, in effect, a second organ in us and influences several risk factors for metabolic syndrome.
- Inflammation, always observed to be a part of the risk factors for metabolic syndrome, may actually be a casual agent, one influenced, in part, by our microbiota.
In Part 2, I’ll dive deeper into how the interplay between microbiota and inflammation may influence the various risk factors contributing to metabolic syndrome, such as obesity, cholesterol and triglycerides.
Stay tuned.
UPDATE: Here’s Part 2, How Gut Bacteria Increase The Chance You’ll Suffer From Metabolic Syndrome And A Simple Solution.
Last Updated on October 16, 2019 by Joe Garma